
Peter Marshall Email and Phone Number
Peter Marshall work email
- Valid
- Valid
- Valid
Peter Marshall personal email
Well-rounded Senior technical leader with 25+ years of experience in product development focused in the medical device industry. Proven track record of successfully bringing innovative products to market in startup, mid-size, and large corporate environments.
-
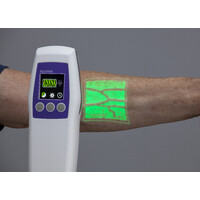
 Vice President Of Research & DevelopmentAccuvein Inc. Oct 2021 - PresentMedford, Ny, UsAccuVein Inc. is the global leader in vein visualization. AccuVein’s flagship product, the AV500, was released in 2019, making it the latest in vein illumination technology. It is built on proprietary technology as embodied in the company’s growing patent portfolio, which currently consists of over 50 issued patents, covering a broad range of imaging and medical diagnostic technologies. AccuVein is now the standard of care in many facilities around the world. To see a demonstration of AccuVein in action, please visit http://www.accuvein.com
Vice President Of Research & DevelopmentAccuvein Inc. Oct 2021 - PresentMedford, Ny, UsAccuVein Inc. is the global leader in vein visualization. AccuVein’s flagship product, the AV500, was released in 2019, making it the latest in vein illumination technology. It is built on proprietary technology as embodied in the company’s growing patent portfolio, which currently consists of over 50 issued patents, covering a broad range of imaging and medical diagnostic technologies. AccuVein is now the standard of care in many facilities around the world. To see a demonstration of AccuVein in action, please visit http://www.accuvein.com -
 Senior Engineering ManagerMedtronic Nov 2007 - Oct 2021Minneapolis, Mn, UsDirectly manage teams of engineers, designers and cross-functional resources; Lead New Product Development activities: design input identification, design verification, clinical evaluation, product validation, and transfer into production; Manage contract design / development and manufacturing activities; Responsible for fiscal year budget planning; Work closely with Senior Management and Business Development to identify strategic opportunities to address gaps in product portfolio, as well as complementary therapies and emerging technologies.Key Accomplishments:• Led the Technical Integration and Remediation of the TruClear product line - inclusive of 4 single use disposable shavers, 3 capital components, and 3 hysteroscopes.• Developed and launched the redesign of single-use hysteroscopic shaver, replacing all machined plastic parts with injection molded components, resulting in a 70% cost reduction.• Performed due diligence for the acquisition of NuWave Surgical’s product line, specifically pertaining to intellectual property, Design History File audits, supply chain and manufacturing capability assessments.• Served as the R&D acquisition integration lead to manage R&D personnel at site, identify and streamline design & manufacturing challenges while successfully transferring line to Covidien facility in Puerto Rico without disruption to supply.• Managed the design and development of a next-generation scope lens cleaner between contract design & development firm and internal resources.• Assumed responsibilities of the Core Team/R&D Lead for Meniscal Repair program to address critical design challenges and manage program to an aggressive 2-year timeline, including supplier development and clinical evaluation protocols, and outsourcing of the production line to a Contract Manufacturer.• Assessed existing product portfolio to identify products to be updated/redesigned, portfolio deficiencies, and products to be classified as obsolete.
Senior Engineering ManagerMedtronic Nov 2007 - Oct 2021Minneapolis, Mn, UsDirectly manage teams of engineers, designers and cross-functional resources; Lead New Product Development activities: design input identification, design verification, clinical evaluation, product validation, and transfer into production; Manage contract design / development and manufacturing activities; Responsible for fiscal year budget planning; Work closely with Senior Management and Business Development to identify strategic opportunities to address gaps in product portfolio, as well as complementary therapies and emerging technologies.Key Accomplishments:• Led the Technical Integration and Remediation of the TruClear product line - inclusive of 4 single use disposable shavers, 3 capital components, and 3 hysteroscopes.• Developed and launched the redesign of single-use hysteroscopic shaver, replacing all machined plastic parts with injection molded components, resulting in a 70% cost reduction.• Performed due diligence for the acquisition of NuWave Surgical’s product line, specifically pertaining to intellectual property, Design History File audits, supply chain and manufacturing capability assessments.• Served as the R&D acquisition integration lead to manage R&D personnel at site, identify and streamline design & manufacturing challenges while successfully transferring line to Covidien facility in Puerto Rico without disruption to supply.• Managed the design and development of a next-generation scope lens cleaner between contract design & development firm and internal resources.• Assumed responsibilities of the Core Team/R&D Lead for Meniscal Repair program to address critical design challenges and manage program to an aggressive 2-year timeline, including supplier development and clinical evaluation protocols, and outsourcing of the production line to a Contract Manufacturer.• Assessed existing product portfolio to identify products to be updated/redesigned, portfolio deficiencies, and products to be classified as obsolete. -
 Director Of Research DevelopmentScandius Biomedical 2001 - 2007UsLead and managed all R&D activities for Class I, II and III medical devices and related instrumentation, coordinated product development projects from the company’s inception through acquisition. Developed schedules, resource allocation plans and budgets for all actively-funded and future projects.Managed all intellectual property initiatives, with aid of outside council, which to date consists of 42 US and international patents and publication across 4 product platforms. Lead innovation, design, and development activities from concept through launch of Stratis® and TriTis® ACL Reconstruction Systems, including design verification, regulatory support, validation studies, vendor qualification, and field training.Implemented and managed design control process meeting FDA and ISO requirements through initial certification and 3 surveillance audits with no reportable actions or findings.
Director Of Research DevelopmentScandius Biomedical 2001 - 2007UsLead and managed all R&D activities for Class I, II and III medical devices and related instrumentation, coordinated product development projects from the company’s inception through acquisition. Developed schedules, resource allocation plans and budgets for all actively-funded and future projects.Managed all intellectual property initiatives, with aid of outside council, which to date consists of 42 US and international patents and publication across 4 product platforms. Lead innovation, design, and development activities from concept through launch of Stratis® and TriTis® ACL Reconstruction Systems, including design verification, regulatory support, validation studies, vendor qualification, and field training.Implemented and managed design control process meeting FDA and ISO requirements through initial certification and 3 surveillance audits with no reportable actions or findings.
Peter Marshall Education Details
-
 Worcester Polytechnic InstituteMechanical Engineering With Biomedical Interests
Worcester Polytechnic InstituteMechanical Engineering With Biomedical Interests
Frequently Asked Questions about Peter Marshall
What company does Peter Marshall work for?
Peter Marshall works for Accuvein Inc.
What is Peter Marshall's role at the current company?
Peter Marshall's current role is Vice President of Research & Development at AccuVein Inc..
What is Peter Marshall's email address?
Peter Marshall's email address is pe****@****nic.com
What schools did Peter Marshall attend?
Peter Marshall attended Worcester Polytechnic Institute.
Free Chrome Extension
Find emails, phones & company data instantly
Aero Online
Your AI prospecting assistant
Select data to include:
0 records × $0.02 per record
Download 750 million emails and 100 million phone numbers
Access emails and phone numbers of over 750 million business users. Instantly download verified profiles using 20+ filters, including location, job title, company, function, and industry.
Start your free trial